Targeted drug delivery technology using untethered microrobots
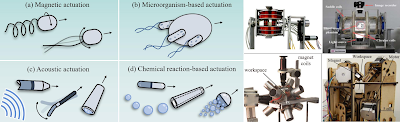
A recent article in the Journal of Micromechanics and Microengineering "Targeted drug delivery technology using untethered microrobots" provided a pretty detailed review of the current mechanisms and structures with cell level treatment delivery option. The article distinguished four typical actuation techniques for in vivo untethered microrobot locomotion with classification according to the actuation energy sources: (a) magnetic actuation; (b) microorganism-based actuation; (c) asouctic actuation; (d) chemical reaction-based actuation.
Abstract
Targeted drug delivery is a promising application of
microrobots owing to the capability of the microrobots to access nearly
every region of the human body through the circulatory system. Research
on microrobots over the past few decades has enabled substantial
advances in the design of both the untethered microrobots swimming in a
biofluid and the related mechanisms to carry and release therapeutic
agents in a controlled manner. This paper presents a comprehensive
review of the technological state of the art in untethered microrobots
for targeted drug delivery applications. First, the in vivo
microrobot locomotion techniques are discussed with respect of the
different types of actuation energy sources such as magnetic fields,
motile microorganisms, acoustic waves, and chemical reaction, outlining
the respective advantages and major limitations. Subsequently, recent
progress in various technologies of microrobot-driven targeted drug
delivery is surveyed deliberating on the corresponding drug manipulation
mechanisms: magnetically driven, motile microorganisms-driven,
acoustic-aided, and stimuli-responsive hydrogels-aided. Although most
studies on microrobot-driven targeted drug delivery were carried out in vitro, few among them successfully demonstrated in vivo
operations in living animals. In the concluding section, current
challenges and future perspectives of the microrobot-driven targeted
drug delivery technology are discussed.
Source: JMM


Comments